 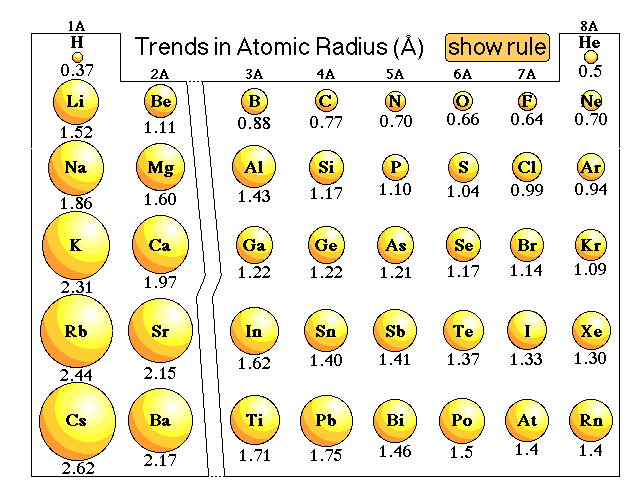
The first ionisation energy is the amount of energy required to remove one electron from the outermost shell of each atom in one mole of atoms of an element in their gaseous state. Periodicity – Trends along Period 3 First Ionisation Energy Trend Across Period 3 Electrical conductivity continues to decrease even further along the period. This is because silicon exists as a giant covalent structure, with no free ions or electrons capable of carrying the current. As we move from aluminium to silicon, electrical conductivity drops dramatically.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
